Matter is a general term for the substance of which all physical objects consist. Typically, matter includes atoms and other particles which have mass. A common way of defining matter is as anything that has mass and occupies volume.In practice however there is no single correct scientific meaning of "matter," as different fields use the term in different and sometimes incompatible ways.

Matter is anything that occupies space or volume and has mass. Matter can be classified in two ways based on physical and chemical properties.
Classification of matter
1. Physical
2. Chemical
Matter is classified into 3 states based on the physical classification.
1. Solid state: Solids state is the state of matter which has definite shape and volume.
2. Liquid state: Liquids have a definite volume but they take the shape of the container ( they dont have a definite shape).
3. Gaseous state: Gases have no definite shape and volume. They take the shape of the container and occupy the volume of the container.
Chemically matter is classified into 3 main groups. They are:-
1. Elements
2. Compounds
3. Mixtures
All these groups are further classified into subtypes (classification of matter)
An element is a pure substance which cannot be further broken down or decomposed into simpler substances by physical or chemical methods.It is made up of atoms with same number of protons. The elements are further subdivided into
1. Metals: Metals are generally solids which have some characteristics like bright luster, good conduction of heat and electricity and hardness. Eg. Na, K, Ca etc.,
2. Non-Metals: Non metals are generally bad conductors of heat and electricity, non lustrous and brittle. Eg. O2, H2 etc.,
3. Metalloids: Metalloids are elements which have common characteristics of both metals and non metals. Eg. Bi, Si etc.,
Compounds are mainly of two types.
1. Organic compounds: Organic compounds are the compounds which are obtained from plant and animal sources.They are the carbon compounds (molecules containing carbon). Eg. Methane
2. Inorganic compounds: Inorganic compounds are compounds obtained from non living sources. Eg. Minerals
A compound is a combination of two or more elements in a definite proportion. The ratio of elements in a particular compound is always same. It can be decomposed into its elements by using some physical or chemical methods. Eg. H2O is a compound where one Oxygen atom combines with two Hydrogen atoms.
Mixture contains a combination of two or more elements or compounds mixed together without any chemical bonds. It can be separated into individual elements or compounds . There are two types of mixtures. They are
1. Homogeneous Mixture: All the components exist in a single phase and they are uniformly mixed. These mixtures are also called as solution.
2. Heterogeneous Mixture: The components exist in more than one phase. They are not uniformly mixed.
Matter exists in one of the three states ; solid, liquid and gas. The state depends upon the temperature and pressure of the substance. The three states are distinguished from one another by differences in macroscopic properties like density, viscosity and elasticity etc., However, from the point of view of atomic models there are only two states with a clear line of demarcation between them. They are ordered state and disordered state. Gases, liquids and some solids called amorphous or glassy solids belong to disordered state. Gases form a dispersed state of matter while liquids and amorphous solids are condensed states of matter. Crystalline solids belong to the ordered state. In practice there may be a little short range order in the disordered structure (liquids) and the ordered state may contain some disorder (imperfections in crystals). This has a significant influence on physical properties of the substancc. It is found that there are some materials with a structure intermediate between the ordered and disordered state. They can be classified as poorly crystalline or as partially crystalline. They include (i) long-chain polymers or plastics (ii) liquid crystals.
Liquid crystals
Liquid crystals are a class of materials intermediate between the disordered state (liquids) and the ordered state (crystals) of matter. They aroused much curiosity in the minds of physicists, chemists and many material scientists because of their electro-optical and other interesting properties. In 1888, Reinitzer observed that when a crystal of an organic substance called cholesteryl benzoate was melted the crystal became a viscous and cloudy liquid at 145.5°C. When the temperature was raised further it became a clear transparent liquid at 178.5°C. When cooled, the opposite transformations occurred at the same temperatures. Lehman further investigated the same substance and observed the following features :
i) It has the ability to flow and low viscosity which are the properties of a liquid.
ii) Exhibits optical anisotropy i.e., birefringence (double refraction) when in the turbid state which is the property of a crystal. Hence, Lehman called this substance in its transition state as liquid crystal. The liquid crystalline phase of the substance is called the 'mesophase'.
The substance having such a character is called a mesogen or liquid crystal.
The liquid crystal appears to resemble a colloidal solution (a suspension of fine particles in water which can be preserved for a long time inspite of their density being different from that of water). However, this is not true because
i) a liquid crystal has well defined and fixed lower and upper temperatures.
ii) the substance undergoes a true phase transition and reveals a distinct phase of matter.
iii) it has orientational order in the mesophase as revealed by X-ray studies.
iv) their molecules are long and rod-like in shape, generally a little flattened, with a typical length of 2.5 nm and a cross-section of 0.6 x 0.4 nm.
v) the molecules are rigid, at least in their central portions.
Thermotropic and lyotropic liquid crystals
The liquid crystals obtained by heating solids i.e., by thermal processes are called thermotropic liquid crystals. The liquid crystals obtained by dissolving certain crystals in suitable solutions are called lyotropic liquid crystals.
The molecules of liquid crystals are elongated and show orientational order leading to particular structures. Based on the structures revealed, Friedel classified liquid crystals into three groups. They are (i) nematic (ii) cholesteric (iii) smectic.
The riematic phase

The nematic phase has the simplest structure. The elongated molecules of the substance are arranged with their axes parallel to each other as shown in Fig.28. 1. Their spatial distribution is completely random i.e., the centres of the molecules are completely random in their location, as in a liquid. In a sheet of perfectly oriented nematic, the molecules have their axes parallel to the walls of the glass slides. They have the same direction every where. The molecules in any layer are parallel to those in the neighbouring layer. The term nematic means 'threadlike' in Greek. When viewed under a powerful microscope, the nematic phase appears as long, thin filaments. The orientational order of molecules is localised. The molecules are capable of moving from one region to another as in a liquid. Each molecule is free to rotate about its own axis. The molecules of true nematic type of liquid crystals are optically inactive. An example of a liquid crystal in the nematic phase is p-azoxyanisole. The temperature range of existence of the phase is 116-136°C.
The cholesteric phase
The cholesteric mesophase is also a nematic type of liquid crystal. It is composed of optically active molecules. The molecules have a rod-like shape-elongated and rigid. They have another very special property, known as chirality. This means that they are asymmetrical. One molecule cannot be made to coincide with its image in a plane mirror by a simple displacement or rotation as shown in Fig. 28.2.
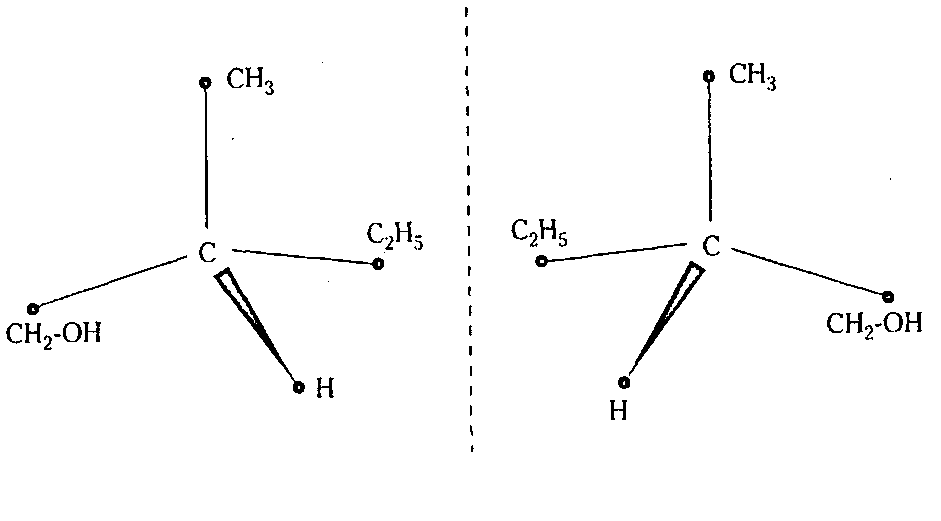
The molecule with such a property is called a chiral molecule. Due to asymmetry, the molecules in one layer make a small angle with those in the adjacent layer. The process is repeated from one layer to another. Consequently the direction of
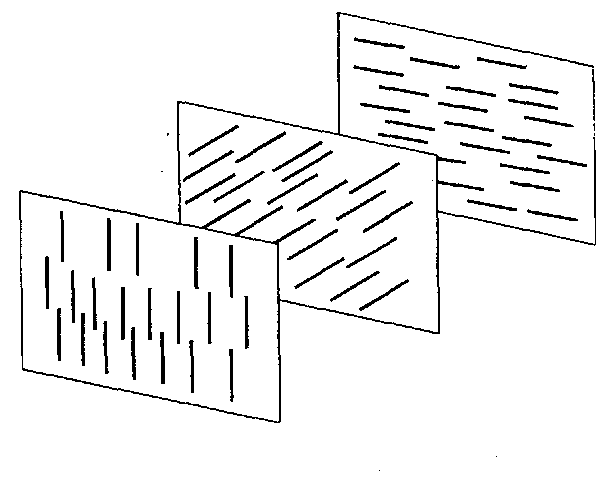
alignment twists progressively, resulting in a helical structure as shown in Fig. 28.3. The molecules recover the same orientation after every 180° rotation. The distance between the two layers in which the molecules have rotated through 180° is called the pitch of the helix. The twist may be right handed or left handed depending on the molecule of the substance. A typical value of the pitch is about 2000 A. This also shows that the cholesteric phase is characterised by a periodicity in a direction normal to the plane containing the axes of the molecules.
The helical structure imparts very special optical properties to the cholesteric phase. Some of them are :
i) the substance exhibits optical activity.
ii) the optical rotatory power (specific rotation) is enormous ; one hundred times greater than that of common optically active solids and liquids.
iii) the colour of reflected light from a sheet of cholesteric material varies with the angle of incidence.
iv) with a given geometry, the colour change varies appreciably with temperature.
Mechanically, a cholesteric liquid has a higher viscosity than a nematic liquid.
An example of a liquid crystal in the cholesteric phase is cholesteryl cinnamate in the temperature range 156 - 197°C
Note :
The name cholesteric originates from the fact that the first examples of this mesophase were encountered in derivatives of cholesterol, a content of blood.
Homework Assignment Help is World No 1 Online Assignment Help Company
@2010 - Copyright © All rights reserved | This is made with by Homework Assignment Help self
In case you face any problem or have any query please email us at :- info[@]homeworkassignmenthelp.com
Submit us an Assignment:
For Demo Class Click hereRead more
Our tutors start working only after the payment is made, to ensure that we are doing work only for serious clients and also our solution meets the required standard.
Getting homework help was never so easy you just need to follow following steps:
(for example: EST, Australian GMT etc)
any example or format you want the solutions to be in.
send you the price quoted by our tutor along with the time needed to solve the assignment
In case you face any problem or have any query please email us at :- info[@]homeworkassignmenthelp.com