Electricity is a general term encompassing a variety of phenomena resulting from the presence and flow of electric charge. These include many easily recognizable phenomena, such as lightning, static electricity, and the flow of electrical current in an electrical wire. In addition, electricity encompasses less familiar concepts such as the electromagnetic field and electromagnetic induction.

Electric charge occurs naturally in materials . electric charge is of two kinds: positive charge and negative charge. For example – when we rub a glass rod with silk cloth , the rod becomes charged. The charge developed on glass rod on rubbing it with silk is called as positive charge. Similarly when an ebonite rod is rubbed with cat-skin, negative charge is developed in ebonite rod. This type of charging of material is called as ‘ charging by friction’.
Atomic Model of electrification
The modern explanation of electrification of material is based on an ‘atomic ‘ or ‘electron model’
According to this model , every material is made up of atoms and atoms are made up of three particles electrons, protons and neutrons. For electrification of a body only electrons are responsible, not the protons because it is not easy to remove the protons from the nucleus.
According to Law of electric charges ‘ Like charges repel each other and unlike charges attract each other’ . For example , a positive charge will attract another negative charge while will repel another positive charge. A negative charge will attract a positive charge and will repel another negative charge.
The force of repulsion or attraction between two like or unlike charges is called as ‘ Coulombian force’.
‘The electric field of a electric charge is the space around the charge in which another charge experiences a force of repulsion or attraction.’
So the intensity of electric field of a electric charge at a point is the ratio of the force acting on the test charge placed at that point to the magnitude of the test-charge. Thus if ‘F’ is the force acting on a test charge ‘q’ placed at a point in an electric field , then the electric field ‘E’ at that point is given by,
E = F /q
Unit of electric field ‘E’ will be ‘Newton per coulomb’.
The electrical conductivity is a term, which is used to describe the ability of the material to pass the electric current through it. The electrical conductivity is due to the movement of charged particles or electrons. The study on the electrical conductivity starts in 18th century by the experiment performed by Benjamin Franklin.The ability of the material to flow the electric current through them is called the electrical conductivity. The electrical conductivity is strongly depends on the temperature of the material.
Classification of materials by conductivity:
A conductor like a metal posses high conductivity and low resistivity.
An insulator like glass possess low conductivity and high resistivity.
The conductivity of a semiconductor is intermediate, but differs under different conditions, like exposure of the material to electric fields or specific frequencies of light, and, also with temperature and composition of the semiconductor material.
The degree of doping in solid state semiconductors produces a large difference in conductivity. The conductivity of a solution of water is highly dependent on its concentration of dissolved salts, and also on other chemical species that ionize in the solution.
Electrical conductivity is the intrinsic property of the material to flow of an electric current through the conductor. Let us consider that a iron rod is attached to a battery and with a galvanometer and we observe that the galvanometer shows deflection. If we use silver rod instead of iron rod we observe that the deflection in the galvanometer is more as compared to the iron bar. That indicates the electrical conductivity of silver is more than the iron. That means every material have their own capacity to conduct the electric current through it. According to the electrical conductivity, silver and aluminum is the best conductors of electric current. If we replace the silver bar by wood or plastics we observe that there is no deflection in the galvanometer. So, that means the wood and plastics do not allow to pass the electric current through them so they are called as the bad conductors of electricity or insulators.
Electrical conductivity is the reciprocal of the resistivity or specific resistance of the material.
Electrical conductivity = 1/ resistivity
The unit of electrical conductivity is mho/metre or siemen/metre or ohm-1 meter-1. Electrical conductivity is the scalar quantity. Electrical conductivity is a temperature dependent quantity. In case of metals electrical conductivity decreases as we increase the temperature of metals. For semiconductors electrical conductivity decreases with increase in temperature. As we decrease the temperature of the metals up to a certain limit, the conductivity becomes too high , this behavior of metal is called superconductivity. :
Introduction to Electric Fields
Electric field is defined as an invisible vector field that exerts force on the charged particles within its vicinity. The concept of an electric field was introduced by Michael Faraday. The electric field is similar to gravitational field with one major difference that it can also exert repulsive forces. The direction of electric field at a point in space is same as the direction of force exerted by the electric field on the test charge. Mathematically speaking, electric field at a point is defined as Lim q -> 0 E = F / q where F is the force exerted on a very small positive test charge q due to the effect of the field E. For example, if a charge of 1 x 10-6 Coulomb experiences a force of 0.2 Newton at at certain point, then the field at that point is 0.2 N / 1 x 10-6 C = 2 x 105 N/C . The SI unit of Electric field is N/C (Newton per Coulomb) or V/m (Volt per meter).
One very important property of Electric fields is the Superposition Principle. It states that if one or more fields is present at a point, then the net field is the vector sum of individual fields. Enet = E1 + E2 + E3 + E4 + ...........
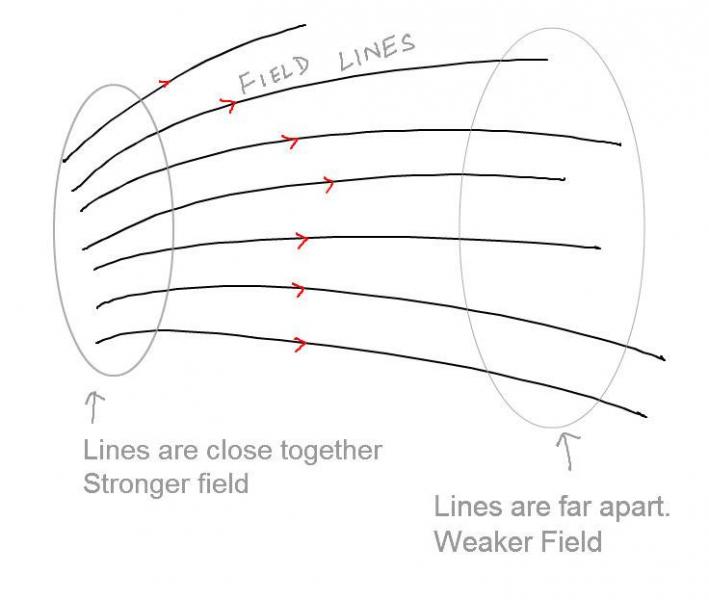
On paper, electric fields (or any such force fields) are represented as directional lines. There are a few rules that one must keep in mind while drawing the electric field.
A sample example of Representation of Electric Fields
Introduction of Electric charge:
Matter is composed of atoms. Electric charge is the basic property of the constituents of an atoms. Each atom contains centrally placed tiny nucleus in which almost the entire mass of it is concentrated. The constituent particles of the nucleus are called nucleons. Nucleons are of two types:
the nucleus is surrounded by electrons. Thus the element of a particle are protons, neutrons and electrons.
Electric charge has been found experimentally that are two types of electric charges:
By convention charge acquired by glass rod rubbed with silk called positive charge; while charge acquired by ebonite rod rubbed with wool called a negative charge.
Proton carries positive charge. The whole positive charge of an atom is due to protons. Every protons are similar. Every protons contain a mass of 1.673 `xx` 10-27kg and one unit of positive electric charge.
Proton charge=+e=+1.6 `xx` 10-19 coulomb.
Neutron is electrically that is it carries no charge. Every neutrons are similar. Every neutron contains a mass of 1.675 `xx` 10-27kg. Neutron charge 0.
It carries negative charge. The whole negative charge of an atom is due to electrons. Every electrons are similar. Each electron has a mass 9.11 `xx` 10-31kg. It is about `(1)/(1836)` of that of a proton. Electron has one unit of negative electric charge. The magnitude of the charge of an electron is almost equal to that of a proton.
Electron charge=-e=-1.6 `xx` 10-19 coulomb.
In normal state and atom is electrically neutral. An atom contains an equal amount of protons and electrons. As charges on proton and electrons. As charges on proton and an electron are equal and opposite so that charge an atom is zero.
Nucleons are bound within nucleus by strong nuclear forces. It is very hard to discard them. However, electrons which move in the outermost orbit of the atom are relatively loosely bound. They can be dislodged by imparting a smaller energy.
An atom gains one unit of positive charge if it is loses one electron. Such an atom called positive ion. If an atom gains one electrons the number of electrons exceeds the number of protons by one. The charge of such an atom becomes negative. Thus an atom acquires one unit of negative charge, such an atom called negative ion.
Homework Assignment Help is World No 1 Online Assignment Help Company
@2010 - Copyright © All rights reserved | This is made with by Homework Assignment Help self
In case you face any problem or have any query please email us at :- info[@]homeworkassignmenthelp.com
Submit us an Assignment:
For Demo Class Click hereRead more
Our tutors start working only after the payment is made, to ensure that we are doing work only for serious clients and also our solution meets the required standard.
Getting homework help was never so easy you just need to follow following steps:
(for example: EST, Australian GMT etc)
any example or format you want the solutions to be in.
send you the price quoted by our tutor along with the time needed to solve the assignment
In case you face any problem or have any query please email us at :- info[@]homeworkassignmenthelp.com